
Just kidding - we're sure you've never heard that joke before. This would allow the swirling to be constant as the burette handler could concentrate on finding the end point accurately.read more.A mole is small, subterranean mammal belonging to the family Talpidae. If the experiment is performed in pairs it would make the burette readings more accurate, as 1 person could swirl the solution and the other could handle the burette. This would make the end point much more accurate. * Add the solution in drips rather than a steady stream.
* Use a thinner burette so that changes in volume are even more noticeable and lowering % error.

This makes the readings the readings more accurate. * Ensure the pipette, burette and volumetric flask readings are done at eye level on a horizontal surface. There are many ways to improve the experiment: * More repetitions, the more repetitions you do the more accurate your end point should be and the average would be more accurate as you are taking into account more results. The % error of the experiment was very small (0.78%) this means the three useable results appear to be reasonably accurate. The average titre was 32.23cm3 therefore the following % error occurred: 0.05/32.23x100= 0.16% There may also be error when using the pipette, if used correctly the error is + or - 0.06cm3 therefore: 0.06/25x100=0.24% The volumetric flask could also produce an error if the bottom of the meniscus rests on the calibration line the error is 0.2cm3.read more. 0.05cm3, this gives an error of + or - 0.05. % Error = actual error/ size of measurement x 100 The balance that was used has an error of + or - 0.005g.
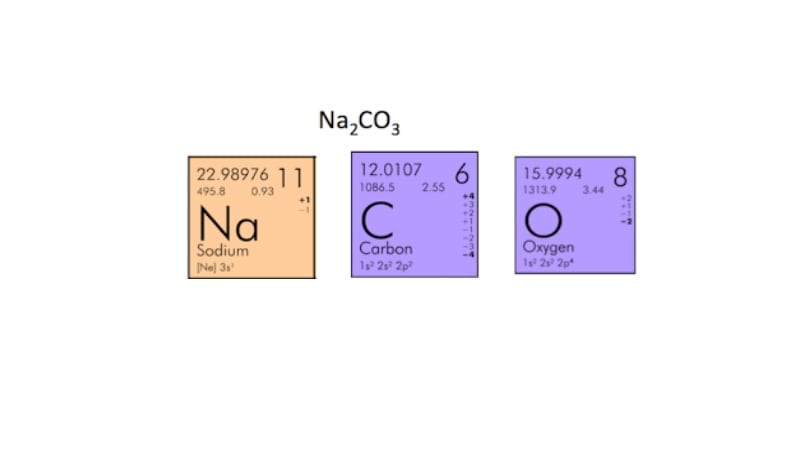
Another factor to consider is percentage error. Out of the 5 results collected 2 have been ignored, this leaves us with only 3 results to find an average with. Or simply missing the end point by not swirling enough or turning off the burette quick enough. There may also have been errors in the readings. Therefore the average is: (31.25+31.20+31.25) / 3 = 31.23cm3 We can now number of moles of calcium carbonate using this equation: Number of moles = concentration x volume 1000 =0.1x25 1000 =0.0025 mol.read more. x 250cm3 1000 Concentration (Na2CO3) = 0.1 mol.dm-3 Results Titration Initial Burette Reading (cm3) Final Burette Reading (cm3) Difference (cm3) 1 (rough) 0.00 32.00 32.00 2 5.00 36.25 31.25 3 1.10 32.30 31.20 4 0.00 31.25 31.25 5 10.00 44.30 34.30 The first and last results are not included in the average since the first is only a rough titration so that later ones can be more accurate and the last is an anomalous result. Finding the concentration of sodium carbonate: Mass (Na2CO3 transferred to volumetric flask) = - =15.24g - 12.59g =2.65g Molar mass (Na2CO3) Na=23 C=12 O=16 2Na x 1C x 3O = molar mass = (2 x 23) + 16 + (3 x 16) =106 Number of mols (Na2CO3) = mass/molar mass =2.65g/106 =0.025mol Number of mols (Na2CO3) = concentration x volume 1000 0.025 mol.
